Abstract
BACKGROUND: Autologous stem cell transplantation (ASCT) for multiple myeloma (MM) entails sudden life changes including acute symptom burden, changes in physical function, and shifting caregiver dynamics. Several studies have shown that anxiety, insomnia, and distress rise in the initial weeks following ASCT before slowly recovering. Long-term consequences of these acute exacerbations include persistent quality of life (QOL) impairments (El-Jawahri 2016), post-traumatic stress disorder (Griffith 2020), and the usage of potentially inappropriate medications (PIMs) for symptom management (Banerjee 2021).
We have recently completed a pilot study of digital life coaching (DLC), whereby life coaches work with patients via phone calls and text messages to provide longitudinal support, education, and accountability to meet wellbeing-related goals. Our pilot study of 15 patients demonstrated the feasibility of DLC during this period, with bidirectional patient-coach engagement occurring every 5-7 days even during index hospitalizations for ASCT (Banerjee 2021). Based on these positive results, we have now launched a randomized Phase 2 study of DLC versus usual care among patients with MM undergoing ASCT.
STUDY DESIGN: Our study is registered at clinicaltrials.gov as NCT04589286. We plan to enroll 60 adult patients with MM undergoing first ASCT at our institution. Inclusion criteria include English language proficiency and ownership of a personal cellphone. However, neither smartphones nor specific mobile apps are required for study participation.
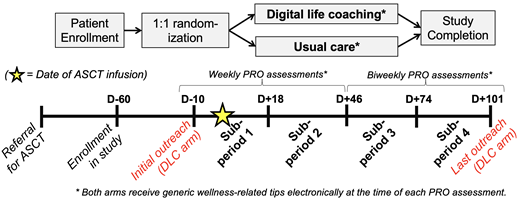
All patients, including those in the control arm, receive brief wellness-related tips with each request for PRO data as outlined below. As shown in the Figure, patients in the DLC arm are paired with a trained life coach beginning at Day -10 before ASCT. Coaches use structured frameworks to assist patients longitudinally with identifying and accomplishing wellbeing-related goals. Specific coaching topics can vary from week to week and are set by each patient. In addition to weekly coach-led phone calls, patients are encouraged to maintain bidirectional communication via phone/text/email as often as desired. Patients in the control arm do not receive access to DLC.
Our primary endpoint is the total usage of sedative-class PIMs - including lorazepam, temazepam, zolpidem, and other similar medications - prescribed for anxiety or insomnia during each of 4 four-week study subperiods identified in the Figure. Secondary endpoints include patient-reported outcome (PRO) assessments of QOL (PROMIS Global Health), distress (NCCN Distress Thermometer), and insomnia (PROMIS Sleep Disturbances 4A). PRO assessments are collected exclusively using automated REDCap emails every 1-2 weeks as shown in the Figure.
PROGRESS TO DATE: As of the data cutoff (7/31/21), 19 patients have enrolled onto our study and 5 have completed all follow-up. The median age of enrolled patients is 62 (range: 31-77), with 26% of patients aged 70 or older. As shown in our pilot study (Banerjee 2021), PRO collection via automated REDCap emails is feasible. Specifically, of 93 email-based requests for PRO assessments as of the data cutoff, 92 (99%) have been completed. Analyses of PRO assessment responses and PIM usage will be conducted after study completion.
DISCUSSION: Improving patient wellbeing during the acute peri-ASCT period is an unmet need in multiple myeloma. Published supportive strategies during this time include music therapy (Bates 2017), acupuncture (Deng 2018), palliative care (El-Jawahri 2017), and programmed hospital room lighting (Valdimarsdottir 2018). DLC may offer unique advantages given its easy accessibility and unified patient-facing interface across hospital/clinic/home transitions. These strengths may be particularly relevant in light of the COVID-19 pandemic, where home-based follow-up after ASCT has become more common. That being said, broadening the accessibility of DLC to include patients with limited English proficiency or patients without personal cell phones are important priorities for future studies.
In summary, our randomized Phase 2 study of DLC versus usual care is ongoing. If shown to reduce PIM prescription rates while improving wellbeing-related PRO trajectories longitudinally, DLC may become a standard of care for patients with hematologic malignancies undergoing ASCT.
Banerjee: Pack Health: Research Funding; SparkCures: Consultancy; Sanofi: Consultancy. Knoche: Amgen: Honoraria. Brassil: Abbvie: Research Funding; Astellas: Research Funding; BMS: Research Funding; Daiichi Sankyo: Research Funding; Genentech: Research Funding; GSK: Research Funding; Sanofi: Research Funding; Pack Health: Current Employment. Jackson: Pack Health: Current Employment. Patel: Pack Health: Current Employment. Lo: Oncopeptides: Consultancy; EUSA Pharma: Consultancy. Chung: Caelum: Research Funding. Wong: Amgen: Consultancy; Genentech: Research Funding; Fortis: Research Funding; Janssen: Research Funding; GloxoSmithKlein: Research Funding; Dren Biosciences: Consultancy; Caelum: Research Funding; BMS: Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees. Wolf: Adaptive Biotechnologies: Consultancy; Teneobio: Consultancy; Sanofi: Consultancy; Amgen: Consultancy. Martin: Oncopeptides: Consultancy; Sanofi: Research Funding; Amgen: Research Funding; Janssen: Research Funding; GlaxoSmithKline: Consultancy. Shah: Bluebird Bio: Research Funding; GSK: Consultancy; Janssen: Research Funding; Indapta Therapeutics: Consultancy; BMS/Celgene: Research Funding; CareDx: Consultancy; CSL Behring: Consultancy; Kite: Consultancy; Nektar: Research Funding; Karyopharm: Consultancy; Amgen: Consultancy; Oncopeptides: Consultancy; Poseida: Research Funding; Precision Biosciences: Research Funding; Sanofi: Consultancy; Sutro Biopharma: Research Funding; Teneobio: Research Funding.